This resource describes:
Which Tau PET tracers have been evaluated in MCI & AD studies?
Although the histopathological hallmarks of Alzheimer’s disease (AD) have long been recognized as the presence of amyloid-beta (Aβ) plaques and neurofibrillary tangles in the brain, it is only recently (2020) that the FDA approved the first Tau PET tracer, [18F]flortaucipir (also known as AV-1451; Tauvid™) to estimate the density and distribution of aggregated tau neurofibrillary tangles in adults with cognitive impairment being evaluated for AD (Barthel, 2020). The main limitations of this ligand include (1) its low sensitivity to earlier tau stages and non-Alzheimer tauopathies, such as corticobasal degeneration (CBD) and progressive supranuclear palsy (PSP) (Soleimani-Meigooni, 2020), and (2) presence of off-target binding, in particular to the enzyme monoamine oxidase B (MAO-B), which may limit routine clinical applications (Leuzy, 2019). Active research is ongoing on second-generation ligands, such as [3H]PI2620, [3H]RO948, [3H]MK-6240, and [3H]JNJ067 (Bischof, 2021; Yap, 2021), which show promising results in overcoming these limitations. A recent comprehensive review of flortaucipir (Burnham, 2024) concluded that, despite its limitations, flortaucipir is an accurate and useful in vivo marker of tau neurofibrillary tangles that can improve the diagnostic, and potentially prognostic, landscape for Alzheimer’s disease. However, using a second-generation ligand ([18F]MK-6240) having a high sensitivity to tau neurofibrillary tangles may allow to detect earlier longitudinal tau deposition in cognitively unimpaired individuals (Pascoal, 2021).

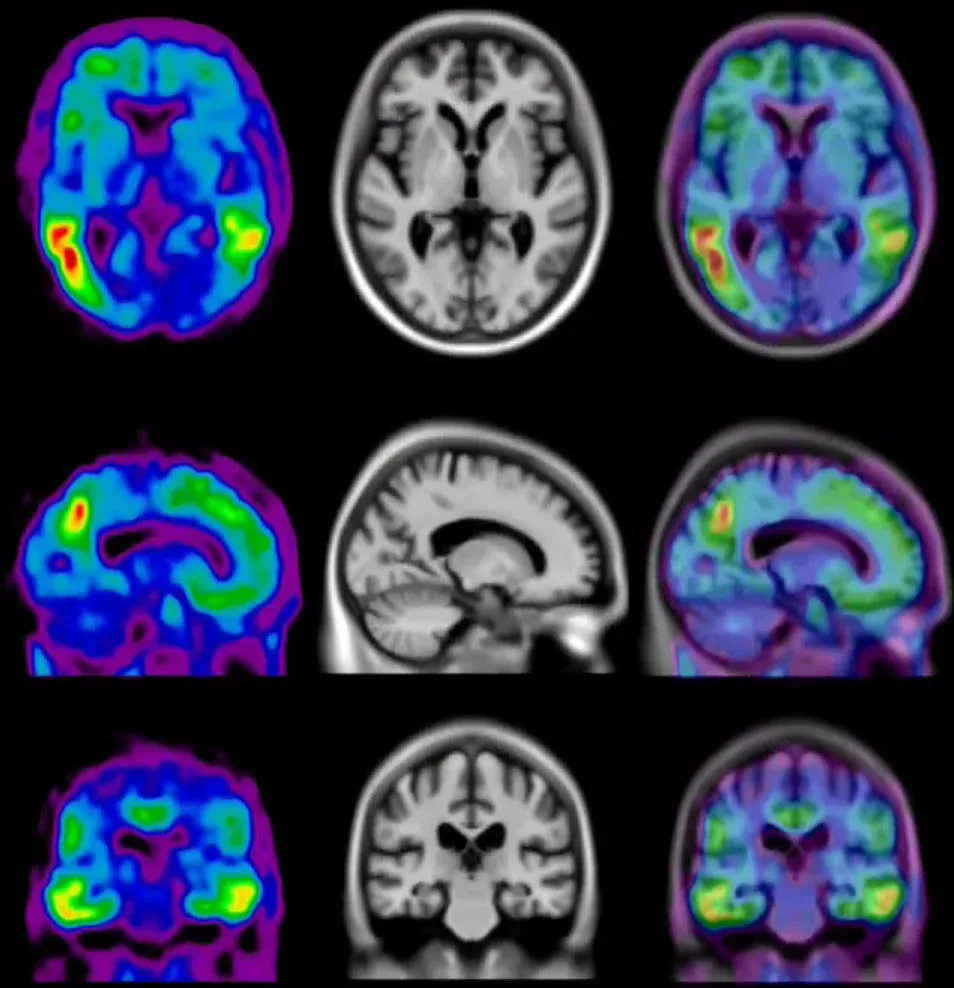
Surface projections of the average Amyloid PET and Tau PET signals observed in a cohort of patients with Alzheimer’s disease. The insets illustrate immunohistochemistry (IHC) staining of an amyloid-beta plaque (left) and intraneuronal tau inclusions & tangles (right).
How does Tau PET compare to other tau biomarkers in predicting disease progression?
Coomans et al. (Coomans, 2023) recently illustrated that Tau PET remains the best predictor of cognitive decline when compared to several other biomarkers, such as plasma tau (pTau181), cortical atrophy on magnetic resonance imaging (MRI), baseline cognition, apolipoprotein E gene (APOE) status, plasma and cerebrospinal fluid (CSF) levels of phosphorylated tau-217, neurofilament light chain (NfL), or amyloid-beta (Aβ)42/40 ratio (Smith, 2023).
However, plasma tau markers might be very helpful in enriching clinical studies (Blennow, 2023). It has been shown that plasma p-tau181 can predict very early AD-related tau aggregation and, as such, may be relevant for trials using tau-targeting drugs given that it creates the possibility of including amyloid-positive, tau-negative individuals at increased risk of imminent tau accumulation in these trials that are designed to halt tau progression at the earliest stages and potentially prevent the subsequent cascade of neurodegeneration and cognitive impairment. Such pre-screening could also reduce the number of patients needed to be enrolled to detect the same drug effect (Ferreira, 2023).
What is the spatiotemporal pattern of Tau PET tracer accumulation?
Pioneering histopathological studies from Braak and colleagues (Braak, 1991) revealed a characteristic deposition pattern of intraneuronal neurofibrillary tangles in AD. A stereotypical progression of pathology, as illustrated in below, occurs from the transentorhinal stages (Braak stages I/II) to a limbic stage (Braak stages III/IV) and finally to a neocortical stage (Braak stages V/VI). [18F]flortaucipir (AV-1451) studies found that an early increased tau deposit in regions of Braak I/II stages with abnormal amyloid-beta status already can identify the individuals whose cognition starts to deteriorate (Chen, 2021). Faster decline in memory performance has been associated with more rapid rates of tau accumulation across the AD spectrum (Jack, 2020). In cognitively unimpaired individuals, the inferior temporal cortex, showed the strongest accumulation over time (Insel, 2023). While the Braak staging system is a good description of tau spreading at the population level, examples have emerged of individual tau patterns that do not fit neatly into this pattern (Ossenkoppele, 2016). As such, a large study of Tau PET-positive individuals revealed that different tau deposition trajectories are associated with different evolution of cognitive impairment, which may be important to consider for the design of future trials (Vogel, 2021).
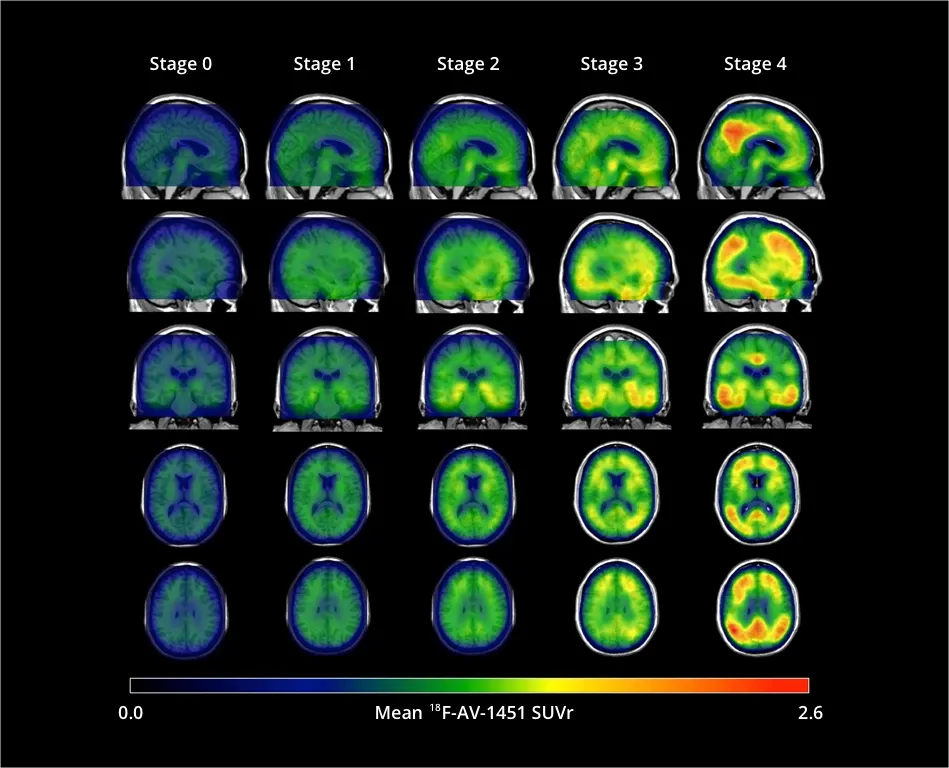
[18F]flortaucipir (AV-1451) signal (standardized uptake value ratio {SUVR]) increases throughout the cortex and subcortex from stage 0 to stage 4. Participants in stage 0 had tau levels corresponding to those of normal young adults. A dominating tau elevation in medial temporal regions (Braak I/II regions of interest) was shown in stage 1. While stage 2 presented increased SUVR in extra-medial temporal regions, stage 3 showed greater SUVR increase in Braak III/IV ROIs including inferior and lateral temporal lobes. Stage 4 had significantly elevated [18F]AV-1451 SUVR extending into the neocortex. Figure reproduced from Chen et al., 2021 under the Creative Commons Attribution License.
The complexity of dissecting the subject-specific regional heterogeneity and longitudinal accumulation of tau may be overcome by using subject-specific regions-of-interest (ROIs), as opposed to using the same ROIs across all subjects for PET tracer analysis. It has been shown that this approach allows for a significant increase in the detection of Tau SUVR annual change for all clinical groups, which resulted in a significant reduction in sample size (Leuzy,2023).
Have changes in Tau PET been shown in clinical trials of MCI/AD?
The historically limited success of amyloid-beta targeting therapies has led to a shift towards the development of tau-targeted therapies. Several potential therapeutic strategies are currently investigated for the treatment of tauopathies, such as antibody-mediated clearance, reduction of gene expression, and small molecule inhibition (Singh, 2024).
The majority of therapeutic agents currently investigated are focused on tau Immunotherapy. Early Phase 2 trials of antibody mediated clearance which included Tau PET as an exploratory endpoint (e.g. zagotenemab, gosuranemab) did not show any significant change in adjusted mean change in the Tau PET standardized uptake value ratio (SUVR) (Fleisher, 2024; Shulman, 2023). One antibody (E2814) demonstrated promising safety, PK, and CSF target engagement profile in Dominantly Inherited Alzheimer’s Disease participants, and is currently being investigated as part of a Phase 3 trial where the primary outcome measure is a change in Tau PET signal (Zhou, 2023).
In terms of gene expression reduction, antisense oligonucleotides (ASOs) may be a promising therapeutic approach. A Phase 1 study using a tau-targeting antisense oligonucleotide (MAPT-RX) showed a dose-dependent reduction in the CSF total-tau concentration (Mummery, 2023), while a Phase 2 study of BIIB080 (MAPT-Rx) is currently underway with preliminary findings showing a reduction of Tau PET signal across all brain regions assessed (Edwards, 2023).
To our knowledge, no clinical trial results using small molecule tau inhibition as a therapeutic agent including Tau PET change as end endpoint have been published (as of September 2024). However, it is likely that the next generation of trials will incorporate Tau PET as is has been deemed critical for biomarker strategies of tau-targeting clinical trials (Penny, 2024).
Our team would be happy to answer any questions about Longitudinal Change in Tau PET in MCI & Alzheimer’s Disease or provide specific information about our other Imaging services.
Discover more about our Imaging Services
Related Content
Up-to-date information on best practices related to the use of neuroimaging in clinical trials of neurological diseases.
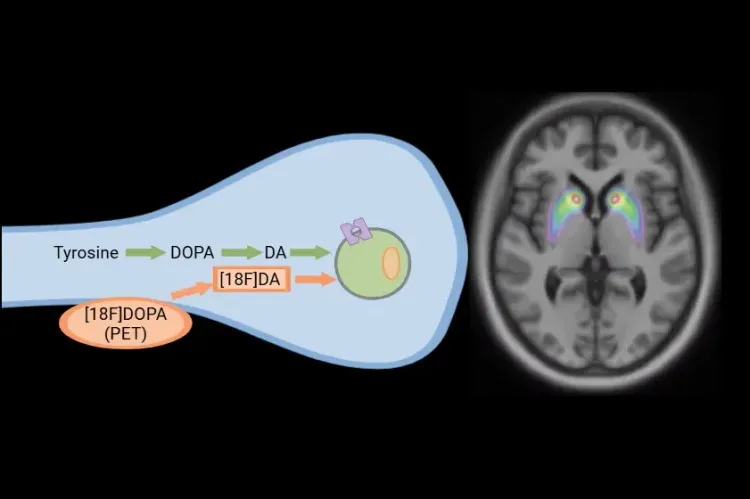
[18F]DOPA PET in Parkinson’s Disease Clinical Trials
How [18F]DOPA PET is used to monitor disease progression & response to therapeutic intervention in Parkinson’s disease & movement disorders clinical trials.
Modeling Protein Misfolding in Neurological Diseases
Computational modeling of protein misfolding mechanisms in neurodegeneration and the application to discovery & development of disease-modifying therapeutics.
Neuroimaging in Frontotemporal Dementia & Clinical Trials
The utility of MRI & PET imaging biomarkers in our understanding of Frontotemporal Dementia (FTD) variants, and their use as endpoints in FTD clinical trials.
MRI Gadolinium-Enhancing Lesions in MS Clinical Trials
How gadolinium (Gd) enhancing lesions are used to track disease progression & response to therapeutic intervention in multiple sclerosis (MS) clinical trials.
Imaging Biomarkers for Progressive Supranuclear Palsy (PSP)
An overview of the various brain imaging methods (MRI, PET, SPECT) available to assess efficacy of disease-modifying therapeutics in clinical trials of PSP.
Imaging Biomarkers to Distinguish CBD from Other Tauopathies
Overview of brain imaging biomarkers to identify Corticobasal Degeneration (CBD) subjects and their use in clinical trials of disease-modifying therapeutics.
MRI in Clinical Trials of Multiple System Atrophy (MSA)
This resource provides an overview of the utility of volumetric MRI and diffusion-weighted imaging (DWI) as biomarkers in research studies of MSA.